Anti-CD38 monoclonal antibodies (mAbs), daratumumab (dara) and isatuximab (isa), are approved to treat multiple myeloma (MM) in the first line and relapsed/refractory settings. Resistance portends a poor prognosis, yet underlying mechanisms have not been well characterized. Despite generally high CD38 expression on most malignant plasma cells (PCs), antigen density may impact the clinical efficacy of targeted therapy. Unfortunately, therapeutic anti-CD38 mAbs interfere with detection and quantification of CD38 surface level by most conventional clinical flow cytometry assays, limiting characterization of CD38 antigen escape.
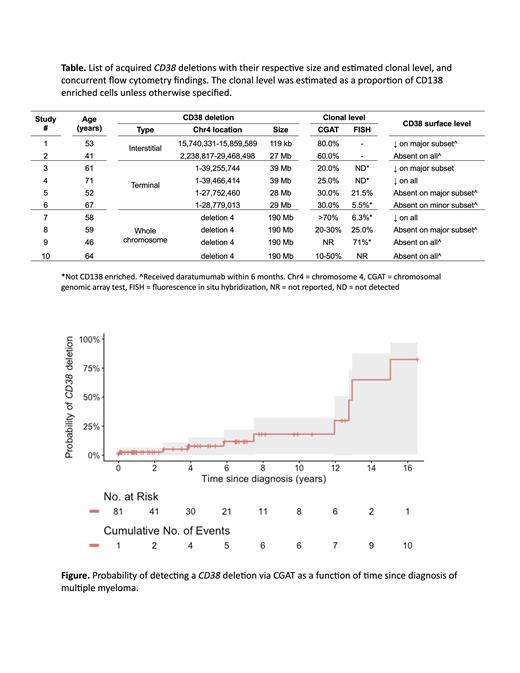
Tumor cell evasion of antigen specific therapeutics can occur with target loss or downmodulation, a phenomenon conventionally referred to as antigen escape. To elucidate mechanisms of CD38 antigen escape, we evaluated MM patients treated with anti-CD38 mAbs who underwent a marrow evaluation between 1/2017 and 10/2022 at a single institution. Eighty-one of 161 (50%) patients had become clinically refractory over their course. Among 82 (51%) patients with ≥1 chromosomal genomic array test (CGAT) performed, 10 (12%) were found to harbor a CD38 deletion ( Table). With a CGAT coverage of 31%, the estimated prevalence of CD38 deletions was 39%. The median interval from diagnosis to CD38 deletion was calculated as 6.7 years (IQR 2.8 to 11.6) for CD38-deleted patients and estimated as 12.9 years (95% CI 12 to NA; Figure) for all patients. The presence of a CD38 deletion was associated with an increasing number of chromosomal abnormalities on concurrent FISH (OR 2.00, 95% CI 1.33 to 3.29, p=0.002).
Specific FISH and flow cytometry findings occurred in conjunction with a CD38 deletion. In 50% of patients, flow cytometry detected a bimodal distribution of CD38 fluorescence intensity, reflecting two populations of malignant plasma cells with distinct CD38 surface levels. No patient showed a uniform malignant plasma cell population with a normal surface CD38 level. When the CD38 (4p15.32) deletion was identified in the context of a large 4p deletion spanning the FGFR3 locus, the FGFR3 (4p16) probe could detect the relevant loss of 4p. This was seen in 6 of 8 patients with a concurrent FGFR3 deletion by CGAT. The proportion of clones harboring a CD38 deletion could be quantified by FISH over time when testing was performed on a CD138-enriched sample.
Low-level CD38 deletions (≤25%) did not confer resistance to dara-containing multi-drug regimens. In two cases where such deletions were present at diagnosis, disease control is ongoing in response to dara-Pd and dara-Rd.
We suggest that acquired CD38 gene deletions appear to be common, the proportion of patients harboring them increases over time irrespective of prior exposure to CD38 mAbs, and their presence facilitates tumor antigen escape. We demonstrate that CGAT can identify CD38 deletions. Conventional clinical flow cytometry assays are unable to quantify the CD38 surface level due to interference from anti-CD38 mAbs. However, we propose that the finding of a bimodal distribution of CD38 fluorescence by flow cytometry could serve as an initial screen to identify at-risk patients warranting confirmatory CD38 mutational analysis. Following characterization of a deletion, FISH could be used to monitor the clonal level longitudinally. Our suggestion that the presence of a CD38 deletion in >25% of malignant PC clones predicts for reduced CD38 mAb efficacy requires prospective validation.
Disclosures
Tuazon:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. Pont:Lyell Immunopharma: Current equity holder in publicly-traded company; Springworks Therapeutics: Consultancy; Cellpoint BV, a Galapagos company: Current Employment, Current equity holder in publicly-traded company. Cole:CellPoint Bio: Current Employment. Newell:Immunoscape: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Neogene Therapuetics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Nanostring Technologies: Membership on an entity's Board of Directors or advisory committees. Green:Janssen Biotech: Consultancy, Research Funding; SpringWorks Therapeutics: Research Funding; Sanofi: Research Funding; Seattle Genetics: Consultancy, Research Funding; Juno Therapeutics A BMS Company: Patents & Royalties, Research Funding; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy; Ensoma: Consultancy; Cellectar Biosciences: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal